Re: Clinical trial cost request, June 2019
In June 2019, KEI reached out to Dr. Jerry Mendell in an effort to obtain information on clinical trial costs.
June 19, 2019 Email from Laurel Boman at KEI to Dr. Jerry Mendell at Nationwide Children’s.
Congratulations on the recent FDA approval of Zolgensma. I am writing to inquire about the clinical trial costs and funding source(s) for trial number NCT02122952, as referenced in the FDA’s approval letter*, or any other related trial.
Knowledge Ecology International (KEI) is among those concerned about the cost of the treatment Zolgensma, and patient access barriers related to the price, not only in the United States, but everywhere. KEI is studying the role of public sector funding for this treatment.
As you may know, the World Health Organization recently adopted a resolution calling for greater transparency around medical technologies, including information on investments, incentives, and subsidies.**
There is currently a lack of information about the costs of clinical trials for the new gene therapies. We do have some information from the California Institute for Regenerative Medicine regarding trial enrollment and costs, but these may or may not be similar to the trials that you have conducted.
We are interested in knowing what was the cost to conduct trials involving Zolgensma, and how much money private charities, such as Sophia’s Cure or Cure SMA, may have contributed, and how much money was invested by the NIH or the private sector.
If you can share information about the costs of trial NCT02122952, or any other SMA gene therapy trials, we would be grateful.
Laurel Boman
James Love
*https://www.fda.gov/media/126130/download
**http://apps.who.int/gb/ebwha/pdf_files/WHA72/A72_ACONF2Rev1-en.pdf
Attachments area
As of August 2, 2019, we have not gotten a response.
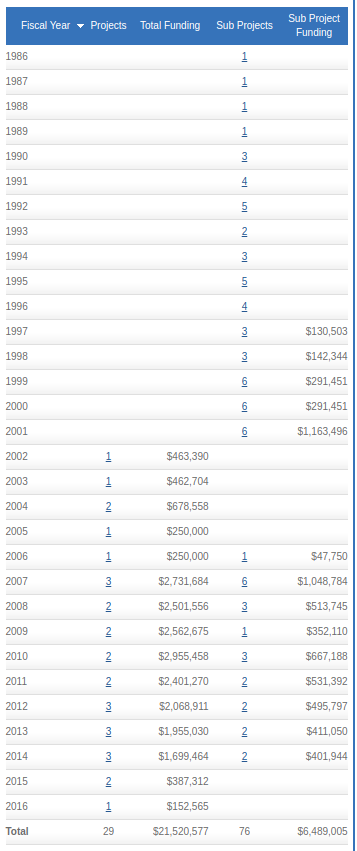
The following are the grant funding in the NIH Reporter database, for Jerry Mendell as a principle investigator.