The WIPO Standing Committee on Copyright and Related Rights is meeting (not as usual) from 12 to 14:30(CET) everyday this week. Day 1 has not gone smoothly because of technological challenges and the Secretariat eventually decided to stop the meeting.… Continue Reading →
On May 8, KEI published KEI Briefing Note: 2026:4, Comments on SCCR/48/3, the March 18, 2026 version of the WIPO Broadcast Treaty Text. KEI-BN-2026-4
On Monday afternoon, 4 May 2026, Knowledge Ecology International (KEI) delivered the following opening statement at the 36th session of the WIPO Committee on Development and Intellectual Property. For development, measures to address access to patented technologies are important, and… Continue Reading →
In 2002, I presented a proposal for a sui generis right for traditional knowledge or access to genetic resources, that would be consistent with more competition and openness. The proposal was modeled after a 1998 EU Directive on Biotechnology, and… Continue Reading →
On Wednesday, 4 February 2026, Knowledge Ecology International (KEI) delivered this constituency statement on behalf of KEI, Health Action International, Global Health Council, World Council of Churches, and Oxfam International: WHO’s work in health emergencies. Our joint statement focused on… Continue Reading →
On Tuesday, 3 February 2026, Knowledge Ecology International (KEI) delivered the following statement on universal health coverage. KEI will make three points. 1. There is inequality in access to small molecule drugs, far more inequality in access to biologic drugs,… Continue Reading →
In a November 25, 2025 decision in the case of
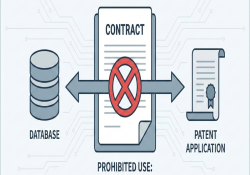
Contracts that restrict patent filing or impose patent-licensing obligations This briefing note illustrates examples of contractual language from data-sharing agreements and material transfer agreements that limit patent filing or impose specific licensing terms. KEI Briefing Note 2025:4 Arianna Schouten December… Continue Reading →
On Tuesday afternoon, 2 December 2025, Knowledge Ecology International made the following statement on copyright limitations and exceptions at the 47th session of the WIPO Standing Committee on Copyright and Related Rights. I wanted to comment first on the African… Continue Reading →
Note on Liability Rules KEI Briefing Note 2025:3 James Love, Thiru Balasubramaniam Draft, October 29, 2025 KEI-BN-2025-3
Selected Documents Prepared by the WIPO Secretariat for the Intergovernmental Committee (IGC) on Intellectual Property and Genetic Resources, Traditional Knowledge and Folklore IGC1 (April 2001) to IGC51 (June 2025) KEI Briefing Note: 2025:2 Arianna Schouten October 28, 2025 KEI-BN-2025-2