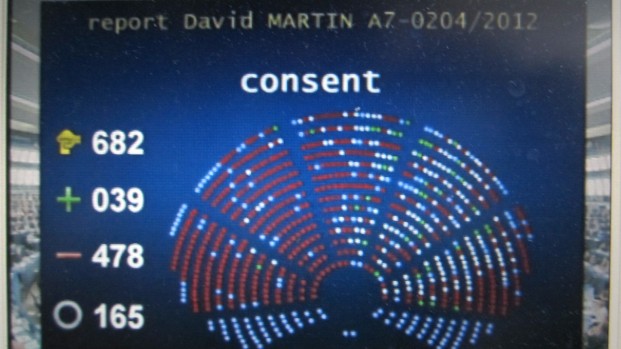
This evening’s informal negotiations at the SCCR 24 on the disabilities issue are over, and delegates are coming out now, with a variety of different stories. Some new document will be tabled Wednesday morning. It will have some important differences, unlike a fairly clean text that was tabled a year ago as SCCR/22/15 REV.1, which was endorsed by Argentina, Australia, Brazil, Chile, Colombia, Ecuador, the European Union and its Member States, Mexico, Norway, Paraguay, the Russian Federation, the United States of America and Uruguay.
Continue Reading →